WHY THIS MATTERS IN BRIEF
Today’s plastics can’t be recycled forever because of the way they’re made and the way recycling systems today work, but a new breakthrough solves both these issues.
 Interested in the Exponential Future? Connect, download a free E-Book, watch a keynote, or browse my blog.
Interested in the Exponential Future? Connect, download a free E-Book, watch a keynote, or browse my blog.
Recycling sounds great in principle, but a frustrating number of devils lurk in the details – and there are plenty of details, from our inability to recycle certain materials through to us just being lazy and not bothering to put our waste in the right trash cans. Similarly, while some materials like aluminum can readily be melted down and turned right back into new aluminum cans over and over again, plastics that are recycled tend to end up being lower quality than the original “virgin” plastic materials that the original product was made from – this is one of the reasons why Adidas had to invent a fundamentally new material for their not quite infinitely recyclable FutureLoop sneakers that I talked about recently.
In the case of plastics though the problem exists because recycled plastic retains some of its previous chemical properties with an analogy being that some of the chemicals within them are like Lego bricks that can’t be separated. And because of this the next plastic product you make out of recycled plastic won’t be exactly the same type or quality as the original, hence the term down-cycling.
Fig 1. Unlike conventional plastics, the monomers of PDK plastic could be recovered and freed from any compounded additives simply by dunking the material in a highly acidic solution. (Credit: Peter Christensen et al./Berkeley Lab). Fig 2. This time-lapse video shows a piece of PDK plastic in acid as it degrades to its molecular building blocks – monomers. The acid helps to break the bonds between the monomers and separate them from the chemical additives that give plastic its look and feel. (Credit: Peter Christensen/ Berkeley Lab). Fig 3. PDK plastics are a “circular” material whose original monomers can be recovered for reuse for as long as possible, or “upcycled” to make a new, higher quality product. (Credit: Peter Christensen et al./Berkeley Lab)
To improve this situation plastics engineers have been trying to create new plastics that can cleanly and easily be broken back down to their original basic components, back to individual Lego bricks that can then be reassembled to create absolutely anything, for some time now. But, the difficulty of accomplishing this feat though is multiplied by all the pigments, flame retardants, and other additives that are used in modern day plastics.
Now though a group led by Peter Christensen at the Lawrence Berkeley National Laboratory in the US have developed a new plastics process that conquers all these challenges to create a new type of plastic that can be infinitely recycled without any deterioration in quality – and that’s a world first.
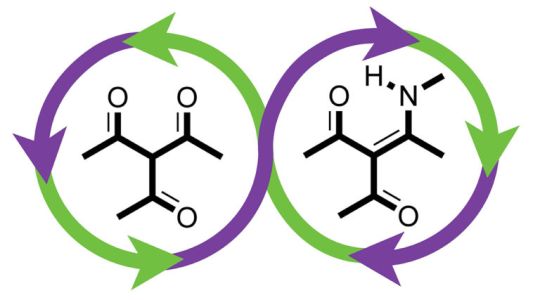
The basic building block of a plastic is called a monomer – connect monomers together and you create polymers with the useful physical properties you’re after. In this case, the researchers are using triketones and amines as building-block monomers, and the process for putting them together sounds like some magic trick, as chemistry often is: combining chemical ingredients causes different building blocks to form, which then spontaneously assemble.
In this case the real trick is how these “diketoenamine bonds” come apart. For most plastics, the energy required to separate the “Lego” blocks is large, making the building process largely irreversible. This is why different types of plastic are generally simply melted down and used to make something of the same type again. But in this case the bonds between the blocks can be easily broken by strong acid, producing monomers that are no different from when you started.
The researchers demonstrated how well the recycling process works with a few examples. First, they coloured pieces of plastic with different pigments, mixing in carbon nanofibers, to boot. After breaking apart the polymer in acid, they were able to easily extract the monomers, leaving behind all the pigments and nanofibers.
In another example, they constructed a flame retardant plastic built on woven fiberglass – like a tarp. Soaking this in strong acid removed the plastic without damaging the fiberglass weave. Another chemical step recovered one pile of pure monomer powder and other piles of the flame retardant additive.
The researchers also tested the physical properties of their “fresh” plastics by remaking them with recycled monomers, and the results were perfect, with no loss of function. They also used recycled monomers to make different plastics, confirming that the building blocks were ready to be something new and could be used to create any other type of products that people might want to create.
The fact that these materials are recycled using strong acids rather than high temperatures even opens up the possibility of extracting them from unsorted collections of plastics, making recycling them en masse an even easier task. And traditional plastics, for better or worse, aren’t affected, so these new infinitely recyclable plastics could be dissolved out and collected while the traditional ones survive intact.
This breakthrough illustrates the possibilities that could help “close the loop” on plastics to create truly circular economy, transforming them into materials with a infinite usable life rather than the single-use, disposable nightmare that we see all too often today.
Source: Nature Chemistry 10.1038/s41557-019-0249-2